One Tiny Drop, One Giant Leap: The Millikan Oil Drop Experiment That Weighed the Unseeable Electron and Unlocked the Quantum World
Discover the electrifying true story of the Millikan Oil Drop Experiment – the 1909 masterpiece that weighed the invisible electron to 0.2% precision. From history and dramatic controversy to modern replications powering your smartphone, AI chips, and 2026 quantum devices, this is the ultimate guide packed with diagrams, real math, mind-blowing applications in nanotechnology, and future tech. Perfect for physics lovers, students, and innovators!
Imagine this: You’re staring through a microscope at a single speck of oil, smaller than a red blood cell, floating in mid-air like a tiny spaceship defying gravity. With a flick of a switch, you make it rise, fall, or hover perfectly still. You’ve just trapped and weighed something no human eye has ever seen – the charge of a single electron.
That’s exactly what Robert Andrews Millikan did in 1909. His oil drop experiment didn’t just measure the tiniest unit of electric charge. It proved electricity is atomic – coming in discrete packets called electrons. It gave us the exact value of e (the elementary charge), a number that underpins every modern device you touch: your phone, laptop, electric car, MRI machine, and the quantum computers racing toward 2026 breakthroughs.
This isn’t dusty textbook history. It’s a living, breathing revolution still shaping nanotechnology, semiconductor chips, and the future of precision engineering. In this epic blog, we’ll travel from Millikan’s Chicago lab in 1909 to today’s cutting-edge labs – with stunning diagrams, step-by-step math, the real controversy, hands-on ideas, and why this experiment is more relevant in 2026 than ever.
Ready to see the invisible? Let’s dive in. (And yes – drop a comment below: What’s the smallest thing you’ve ever measured?)
ROBERT ANDREWS MILLIKAN/n(1868-1953). American physicist. Robert Andrews Millikan (1868–1953) – the man who caught electrons one drop at a time. (Historical portrait, c. 1930)
1. The Quest for the Electron: Why the World Needed This Experiment (Pre-1909 History)
Electricity has fascinated humans for millennia – from ancient amber attracting feathers to Benjamin Franklin’s kite in 1752 proving lightning is electric. By the late 1800s, scientists knew electricity involved tiny particles, but how tiny? And was charge continuous like water… or chunky like atoms?
J.J. Thomson’s 1897 cathode-ray experiments showed negatively charged “corpuscles” (later called electrons) with a charge-to-mass ratio, but no one knew the actual charge e or mass m separately. Estimates varied wildly – from 10^{-10} to 10^{-20} coulombs. Without e, you couldn’t calculate Avogadro’s number, Planck’s constant, or build reliable electronics.
Enter Robert Millikan, a meticulous physicist at the University of Chicago. Working with student Harvey Fletcher, he asked the ultimate question: Is electric charge quantized? If yes, then electrons are real fundamental particles. If no, maybe charge is continuous.
Think about it: If you could measure the charge on hundreds of tiny objects and always found it as 1×, 2×, or 3× some base value (never 1.37×), you’d prove nature’s graininess at the smallest scale. That’s exactly what Millikan did.
His innovation? Use oil instead of water (no evaporation), ionize with X-rays for controlled charging, and balance gravity with electric force on individual drops. Genius.
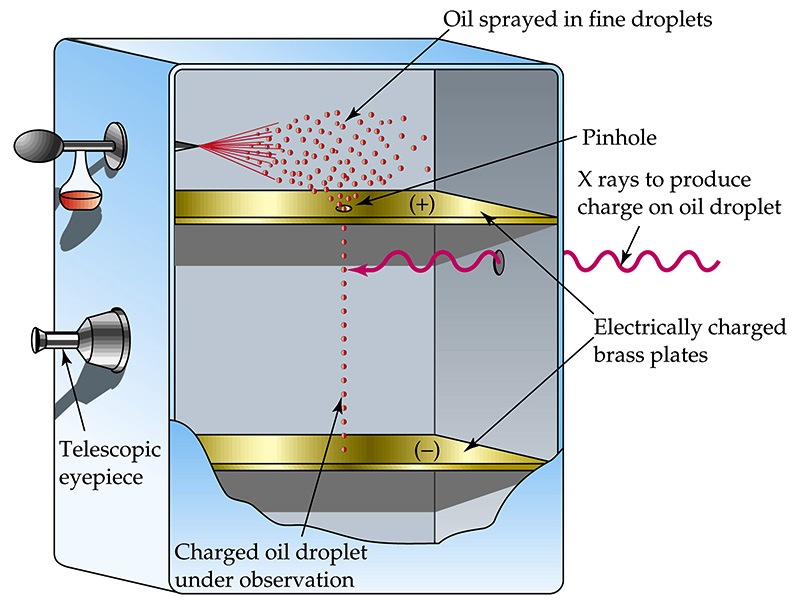
Classic schematic of the Millikan apparatus – oil mist, charged plates, microscope view. (Modern educational diagram)
2. The Apparatus: A Precision Stage for the Invisible (The Setup Explained)
Picture a sealed chamber between two parallel brass plates, 16mm apart, connected to a high-voltage battery (up to 10,000 V). A tiny pinhole in the top plate lets oil droplets (1–10micrometers diameter, like fog particles) drift in from an atomizer. Illumination comes from a bright lamp at 90° angle – drops sparkle like stars. You observe them through a microscope with a scale, timing their fall and rise. X-rays ionize the air, so drops pick up or lose electrons one at a time.
Key forces on each drop:
- Gravity (mg): downward
- Buoyancy (small correction)
- Viscous drag (Stokes’ law: 6πηrv, where η is air viscosity, r radius, v velocity)
- Electric force (qE): upward or downward depending on charge sign and field direction
When the electric field is off, the drop falls at terminal velocity v₁. When on, it rises at v₂. At balance (stationary), qE = mg.
Millikan measured velocities to 0.01seconds accuracy over tiny distances. He repeated on the same drop for hours, changing its charge dozens of times.
The magic equation (simplified):
The charge q on a drop is: (where ρ is oil density, E is electric field = V/d).
But first you find radius r from fall speed using Stokes’ law (with corrections for tiny drops). Every q turned out to be n × e, where n is a small integer.
Interactive moment: Grab a calculator. If a drop falls at 0.001cm/s and rises at 0.002cm/s under known voltage, what’s q? (We’ll solve real examples later!)
Detailed forces diagram – without field (falling) vs. with field (rising). Buoyancy, drag, weight, electric force in perfect balance.
Full lab setup photos and modern replica – atomizer, plates, microscope, stopwatch. Real 1909–1913 equipment looked almost identical!
3. The Breakthrough: How Millikan Proved Charge is Quantized (Results & Math Deep Dive)
Between 1909 and 1913, Millikan observed hundreds of drops. In his landmark 1913 Physical Review paper, he reported 58 pristine ones. Every charge was an integer multiple of e = 1.592 × 10-19C (his value; today’s exact value is 1.602176634 × 10-19C).
Real data example from Millikan’s notebooks (simplified):
Drop A: charge = 4.8 × 10-19C → 3e
Drop B: 3.2 × 10-19 C → 2e
Drop C: 1.6 × 10-19 C → 1e
Full derivation (for physics lovers):
Terminal velocity without field:
(σ = air density). Solve for r.
With field:
Combine and solve for q. Millikan’s genius was observing the same drop changing charge (by ±e, ±2e, etc.) while keeping radius constant. His final value had just 0.2% uncertainty – revolutionary. It matched Thomson’s e/m ratio perfectly, giving electron mass m_e ≈ 9.11 × 10-31kg.
Mind-blowing stat: One electron’s charge is so small that 1 ampere (everyday current) is ~6.24 × 1018 electrons per second. That’s 6 quintillion!
The Drama & Controversy: Data Selection, Ehrenhaft, and “Fraud?” Claims
No great scientific story is without tension. Felix Ehrenhaft in Vienna claimed “sub-electrons” – charges smaller than e. He used similar methods but got different results.
Millikan’s notebooks (now public) show he discarded ~100 of ~175 drops – those too big/small, Brownian-motion affected, or inconsistent. In his 1913 paper he wrote: “This is not a selected group of drops…” – a statement later called misleading.
Was it fraud? Historians (Allan Franklin, Gerald Holton, David Goodstein) say no. Millikan was perfecting technique in early runs. Including bad data would have worsened accuracy. Reanalysis of all his data changes e by <0.1%. He was seeking truth, not faking it.
This debate teaches real science: data selection is normal when justified. Textbooks often skip this – but understanding it makes you a better scientist.
Question for you: If you had noisy data, would you publish everything or select the cleanest? Comment below!
5. Nobel Glory & Immediate Impact (1923 Onward)
Millikan won the 1923 Nobel Prize in Physics “for his work on the elementary charge of electricity and on the photoelectric effect.” (He also measured Planck’s constant accurately.)
His e value became the bedrock of physics.
It enabled:
- Accurate Avogadro’s number
- Better atomic models
- Early electronics design
Discovery of the Parts of the Atom: Electrons and Nuclei – Millikan Oil Drop Experiment and Rutherford Scattering (Millikan receiving his Nobel – the ultimate validation of one drop at a time.)
6. Modern Replications: Doing It Better in 2026 Labs
Today’s undergrad labs use digital cameras, CCD sensors, and automated tracking. Precision is now 0.01% or better. Some use laser trapping or single-electron transistors.
Recent papers (2020s) confirm e to parts-per-billion using quantum metrology. The 2019 SI redefinition fixed e exactly – thanks to Millikan’s foundation!
7. Present-Day Power: From Your Phone to Nanotechnology & Semiconductors
Every transistor in your device relies on electron charge control. In nanotechnology:
- Atomic Layer Deposition (ALD) builds chips atom-by-atom – precision traceable to e.
- Quantum dots and single-electron transistors use quantized charge for ultra-low-power computing.
- Medical nanosensors detect single-electron events for cancer diagnostics.
2026 relevance: AI chips need atomic precision. Millikan’s e helps calibrate them.
 Nanoscale transistors could enable more efficient electronics | MIT News | Massachusetts Institute of Technology
Nanoscale transistors could enable more efficient electronics | MIT News | Massachusetts Institute of TechnologyNanoscale transistors – the direct descendants of Millikan’s discovery.
8. Future Frontiers: Quantum Computing, Precision Metrology & Beyond 2026
In 2026, single-electron pumps define the ampere. Quantum computers use electron spin/charge qubits. Ultrafast electron microscopy images charge dynamics at femtosecond scales.
Innovative vision: Imagine “Millikan 2.0” – trapping single atoms with optical tweezers for even finer constants. Or using ALD to build custom quantum devices where electron charge is engineered on demand. The experiment that started with oil drops now powers the quantum revolution.
9. Try It Yourself: Home/School Versions & Thought Experiments
Affordable kits exist (~$200). Or simulate online (PhET or ComPADRE).
Simple thought experiment: If electrons had fractional charge, your phone battery would behave differently. What would change?
10. Fun Facts & Mind-Blowing Stats
- One oil drop holds ~10–100 electrons.
- Millikan observed drops for hours.
- e is now exact – no uncertainty!
- The experiment inspired Feynman’s “atomic hypothesis.”
Conclusion: The Legacy That Keeps Electrifying the Future
One tiny drop. One giant proof that the universe is quantized. Millikan’s experiment didn’t just measure charge – it revealed nature’s fundamental graininess and gave us the tools to engineer the 21st century.
In 2026, as we build 2D materials, quantum devices, and AI hardware atom-by-atom, we stand on his shoulders.
What surprised you most? How does knowing this change how you see everyday tech? Share this post, comment your thoughts, and tag a physics friend. Let’s keep the conversation alive – because the next breakthrough might start with your question!
References & Further Reading
- Millikan, R.A. (1913). Physical Review.
- Millikan’s Nobel Lecture (nobelprize.org).
- Weinberg, S. (1983). The Discovery of Subatomic Particles.
- Goodstein, D. (defense of data selection).
- Modern texts: University Physics by Young & Freedman.
- Recent replications: IOP Science & NIST papers on single-electron metrology.



Comments
Post a Comment